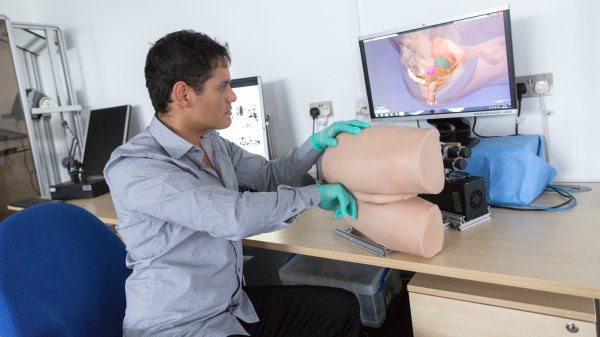
One of our primary goals here at the International Prostate Cancer Foundation is to be on the forefront of both research and education. Which mean we are always looking to the newest innovations in prostate cancer research and treatment. This year there have been many strides made in cancer detection. These are just a few of some of the amazing advancements moving the cause forward.
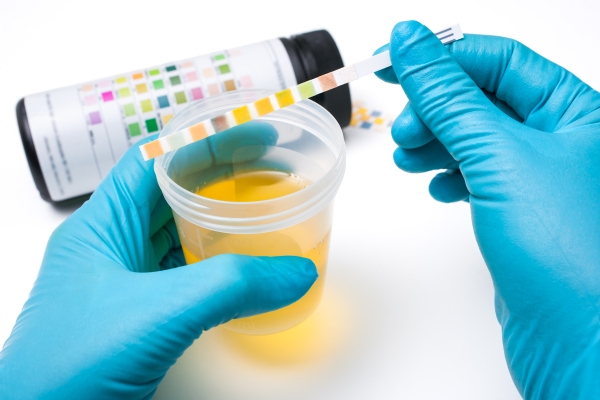
Testing for non-coding RNA molecules in urine may offer a way to detect prostate cancer that is more accurate and reliable than current methods using biomarkers such as PSA and PCA3.
This was the conclusion of a German study presented at the European Association of Urology Congress (EAU16) in Munich, Germany, March 11-15, 2016.
Friedemann Horn, a professor in the University of Leipzig and the Fraunhofer Institute for Cell Therapy and Immunology IZI, and Manfred Wirth, a professor in the University of Dresden – both in Germany – led the work.
Prof. Wirth says:
“Our work on RNAs [ribonucleic acid] is allowing us to design a completely new kind of prostate cancer test.”
Current biomarker tests for prostate cancer measure levels of PSA (prostate-specific antigen) and PCA3 (prostate cancer gene 3), but they are not particularly accurate and can either miss many cancers or produce false positives.
The U.S. Food and Drug Administration has approved Axumin, a radioactive diagnostic agent for injection. Axumin is indicated for positron emission tomography (PET) imaging in men with suspected prostate cancer recurrence based on elevated prostate specific antigen (PSA) levels following prior treatment.
Prostate cancer is the second leading cause of death from cancer in U.S. men. In patients with suspected cancer recurrence after primary treatment, accurate staging is an important objective in improving management and outcomes.
“Imaging tests are not able to determine the location of the recurrent prostate cancer when the PSA is at very low levels,” said Libero Marzella, M.D., Ph.D., director of the Division of Medical Imaging Products in the FDA’s Center for Drug Evaluation and Research. “Axumin is shown to provide another accurate imaging approach for these patients.”
Two studies evaluated the safety and efficacy of Axumin for imaging prostate cancer in patients with recurrent disease. The first compared 105 Axumin scans in men with suspected recurrence of prostate cancer to the histopathology (the study of tissue changes caused by disease) obtained by prostate biopsy and by biopsies of suspicious imaged lesions. Radiologists onsite read the scans initially; subsequently, three independent radiologists read the same scans in a blinded study.
The second study evaluated the agreement between 96 Axumin and C11 choline (an approved PET scan imaging test) scans in patients with median PSA values of 1.44 ng/mL. Radiologists on-site read the scans, and the same three independent radiologists who read the scans in the first study read the Axumin scans in this second blinded study. The results of the independent scan readings were generally consistent with one another, and confirmed the results of the onsite scan readings. Both studies supported the safety and efficacy of Axumin for imaging prostate cancer in men with elevated PSA levels following prior treatment.
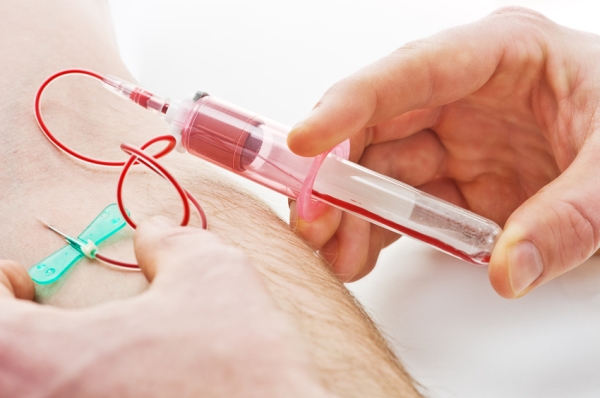
A new UBC-developed method to isolate cancer cells that have escaped from a tumour could soon pave the way for improved diagnosis and treatment.
The simple process involves a special device that squeezes cells in a blood sample through tiny funnels, which drive the cancer cells and blood cells into separate streams based on differences in their size and softness.
“Circulating tumour cells–cells from a tumour that have escaped into the bloodstream with the potential to spread into other tissues–are extremely useful for assessing a patient’s disease in order to select the most appropriate treatment,” said UBC mechanical engineering professor Hongshen Ma, the lead researcher. “These cells are particularly important for prostate cancer, where the site of metastasis is typically in the bone, where biopsies are difficult or impossible.”
Ma’s research focus is microfluidics, the flow of liquids through channels smaller than a human hair. The microfluidic device designed by his team captures cells based on their distinct internal structure–a mechanical analysis instead of the blood chemistry analysis used in conventional medical diagnostic techniques.
The device was first tested using blood samples spiked with cancer cells. It was then used to analyze blood samples from 20 patients with metastatic castration-resistant prostate cancer, an advanced form of cancer, and from four healthy individuals.









 In 2012, the task force
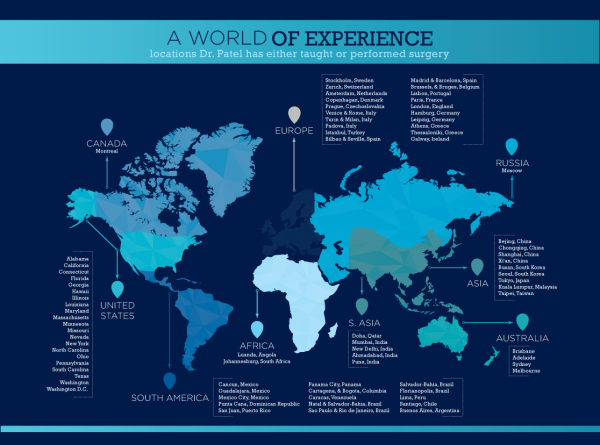
In 2012, the task force If you follow us, you know that our founder Dr. Vipul Patel is out to change the world!! Not only does he do amazing work here in the United States, but he is also always traveling the world to educate and train other physicians and patients.
If you follow us, you know that our founder Dr. Vipul Patel is out to change the world!! Not only does he do amazing work here in the United States, but he is also always traveling the world to educate and train other physicians and patients.